(Read May 23, 1901.)
In a recent number of the Comptes Rendus, an account was given by Curie of the evidence of the existence of a new gas from radium, which possesses remarkable physical properties. A specimen of very radioactive radium was placed in a glass vessel connected with a mercury pump. On exhausting to a low vacuum and allowing the apparatus to stand, the pressure steadily increased. When the very small volume of gas thus collected flowed along the glass tubes, it rendered them phosphorescent, and if left in for some time, rapidly blackened them. The gas itself was powerfully radioactive, i.e. it continuously gave out a type of Röntgen rays, which made gases partial conductors of electricity and rapidly acted on a photographic plate. This gas preserves its radioactive power for several weeks.
For some time past, one of the authors had been independently investigating one of the most remarkable properties of radioactive substances, namely, the power of continuously emitting radioactive particles of some kind. The term "emanation" was applied to the substance thus emitted, as there was no evidence at the time whether the material emission was a vapour of the substance, a radioactive gas, or particles of matter each containing a large number of molecules.
The "emanation" from thorium compounds was shown to retain its radioactivity for several minutes and possessed the remarkable property of causing every substance in the neighbourhood of the thorium to become itself radioactive for several days. The specimens of impure radium then in the possession of the author, did not possess the power of emitting such an emanation; but Dorn, using a later and more active preparation of radium, showed that it possessed the same emanating power as thorium. One of the most interesting properties of excited radioactivity is that it can be concentrated in an electric field on the kathode, so that a very fine wire of any metal can be made to act like a powerfully radioactive substance for several days.
A short time ago, one of the authors published an account of the effect of temperature on the emanating power of radioactive substances, in the Physikalische Zeitschrift. In the paper it was shown that the emanating power of thorium increased with rise of temperature to about a red heat, but on heating to a white heat the emanating power was destroyed and could not be recovered. An examination of a specimen of radium obtained from De Haen, Hanover, showed that the effect of temperature on its emanating power was very large. When the substance was heated below a red heat, its emanating power increased over 10,000 times, but was to a large extent destroyed by heating to a higher temperature. The emanation, obtained by passing a slow current of air over heated radium, was found to preserve its radioactive powers for weeks, when kept in a closed metal vessel. The radioactivity slowly decayed, but was still quite appreciable after a month's interval.
The question now arose, if any physical experiments could be devised to settle the problem as to whether the emanation was in reality a radioactive gas, driven off from the substance, or a vapour of the substance, or a material emission of particles much larger than molecules. Experiments on thorium showed that no appreciable volume of gas could be collected by leaving thorium oxide in a vacuum tube connected with a mercury pump. No new lines were observed in the spectrum of the gas. The amount of the emanation thus given off was thus too small to detect by its volume in this way, but the electrical conductivity produced by the emanation in the gas, with which it is mixed, is often very large and can be used as a measure of the amount of emanation present. The emanation gives out a type of radiation which ionizes the surrounding gas. When a strong electric field is applied, the current through the gas reaches a maximum value, and is then a measure of the total number of ions produced per second, multiplied by the charge on the ion.
By determining the rate of diffusion of the emanation into air or
other gases, using the electrical method, it is possible to obtain
an approximate estimate of its molecular weight. The coefficient of
inter-diffusion of most of the simple gases have long been known, and
the results show the coefficient of diffusion of one gas into another
is inversely proportional to the square root of the product of the
molecular weights. If therefore the coefficients of diffusion of the
emanation into air is found to have a value lying between that of two
gases and
, we can conclude that the molecular weight of the
emanation lies between the molecular weights of
and
.
The apparatus employed was similar to that used by Loschmidt[1] in his experiments on the coefficient of inter-diffusion of gases in the year 1871.
Fig. (1) shows the general arrangement. A long brass cylinder
6 cms . in diameter, 73 cms. long, was divided into two equal parts
by a movable metal slide
. The ends of the cylinder were closed
with ebonite stoppers. Two insulated brass rods
and
, each
half the length of the tube, passed through the ebonite stoppers and
were supported centrally in the tube. The cylinder was insulated and
connected to one pole of a battery of 300 volts, the other pole of
which was to earth. The central rods could be connected to a sensitive
quadrant electrometer.
The cylinder was covered with a thick layer of felt, and placed inside a metal box filled with cotton wool, in order to keep temperature conditions as steady as possible.
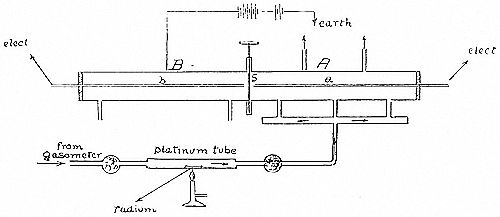
Fig. I.
In order to carry a sufficient quantity of emanation into the half
cylinder , it was necessary to slightly heat the radium. The slide
was closed and the side tubes opened. A slow current of dry
air from a gas bag, passed through a platinum tube, in which a small
quantity of a radium compound was placed. The emanation was carried
with the air into the cylinder
. When a sufficient quantity had
been introduced, as tested by the electrometer, the current of air was
stopped. The side tubes were closed by fine capillary tubes. These
prevented any appreciable loss of gas due to diffusion, but served
to keep pressure of gas inside
at pressure of outside air. The
three entrance tubes into the cylinder, shown in the figure, were for
the purpose of initially mixing the emanation and gas as uniformly as
possible.
After standing for several hours to make temperature conditions steady,
the slide was opened, and the emanation began to diffuse into the tube
.
The current through the tubes and
was measured by an
electrometer, with suitable capacity in parallel, at regular intervals.
Initially there is no current in
, but after the opening of the
slide, the amount in
decreased and the amount in
steadily
increased. After several hours the amount in each half is nearly
the same, showing that the emanation is nearly uniformly diffused
throughout the cylinder.
It can be readily shown that if
See Stefan and Loschmidt, Berichte Wien. Akad., 63, 1871.
From this equation can be determined, if
and
are known.
An uncertainty however arises in estimating and
for the rate of leak in
and
is made up of the current due
to emanation alone and the current produced in the gas by the excited
radioactivity on the electrodes. As the amount of excited radioactivity
increases with the time, the ratio of the current due to the emanation
and to the excited radiation varies with the time allowed for
diffusion. The ratio of the current due to the excited radiation can be
determined by removing the central electrode and finding the amount of
current immediately after the introduction of a new electrode.
When the emanation is allowed to diffuse for half an hour, the current
due to excited radioactivity was about of the whole.
The calculated value of was found to be about 20 per cent greater
when the correction for the amount of excited radioactivity was applied.
The value of deduced from the experiments was found to be between
and
. All the later observations gave a value
about
.
This variation in the value of deduced from the experiments is
not altogether due to errors of experiment, the values obtained at
first with a new specimen of radium were in all cases higher than when
it had been laid by for several months. It appears as if the emanation
were not simple in character, and that part of the emanation first
given off was of lower molecular weight than that emitted after several
months exposure to the air. Further experiments are now being carried
out to see if the radium emanation undergoes a progressive change with
time. For the purpose of comparison, we will now give a few of the
coefficients of inter-diffusion of gases, compiled from Landölt and
Bernstein's tables.
| Gas or vapour | Coefficient of diffusion into air. | Molecular weight |
|---|---|---|
| Water vapour | 18 | |
| Carbonic acid gas | 44 | |
| Alcohol | 46 | |
| Ether | 74 |
In the above table we see that the coefficient of inter-diffusion
follows the inverse order of the molecular weights. In cases of the
simpler gases it has been shown experimentally that the coefficient
of inter-diffusion is approximately inversely proportional to the
square root of the product of the molecular weight. If we apply these
considerations to the emanations
we see that it is a gas or a vapour of molecular weight (allowing
a wide margin) probably lying between 40 and 100. These numbers exclude
the possibility of the substance being a vapour of radium, for it has
already been shown by M. and Mme. Curie that the atomic weight of
radium is greater than that of barium. We must therefore conclude that
the emanation is in reality a heavy radioactive vapour or gas.
On account of the rapid decay of the radiating power of thorium emanations, it is not possible to determine its coefficients of diffusion in the same way; but special experiments show that it diffuses rapidly, and is also probably gaseous in character. T he physical properties of these emanations or gases are most remarkable. The radium emanation not only continues for long inter vals to be a source of radiation which is apparently similar in character to easily absorbed Röntgen rays, but in some way manufactures from itself a positively charged substance, which travels to the negative electrode and becomes a source of secondary radioactivity.
[1] Wiener Akad., 1871.
TRANSCRIBER’S NOTES
This article was published in the Proceedings And Transactions Of The Royal Society Of Canada, Second Series, Volume VII, in the Meeting of May, pp. 21-25, 1901.